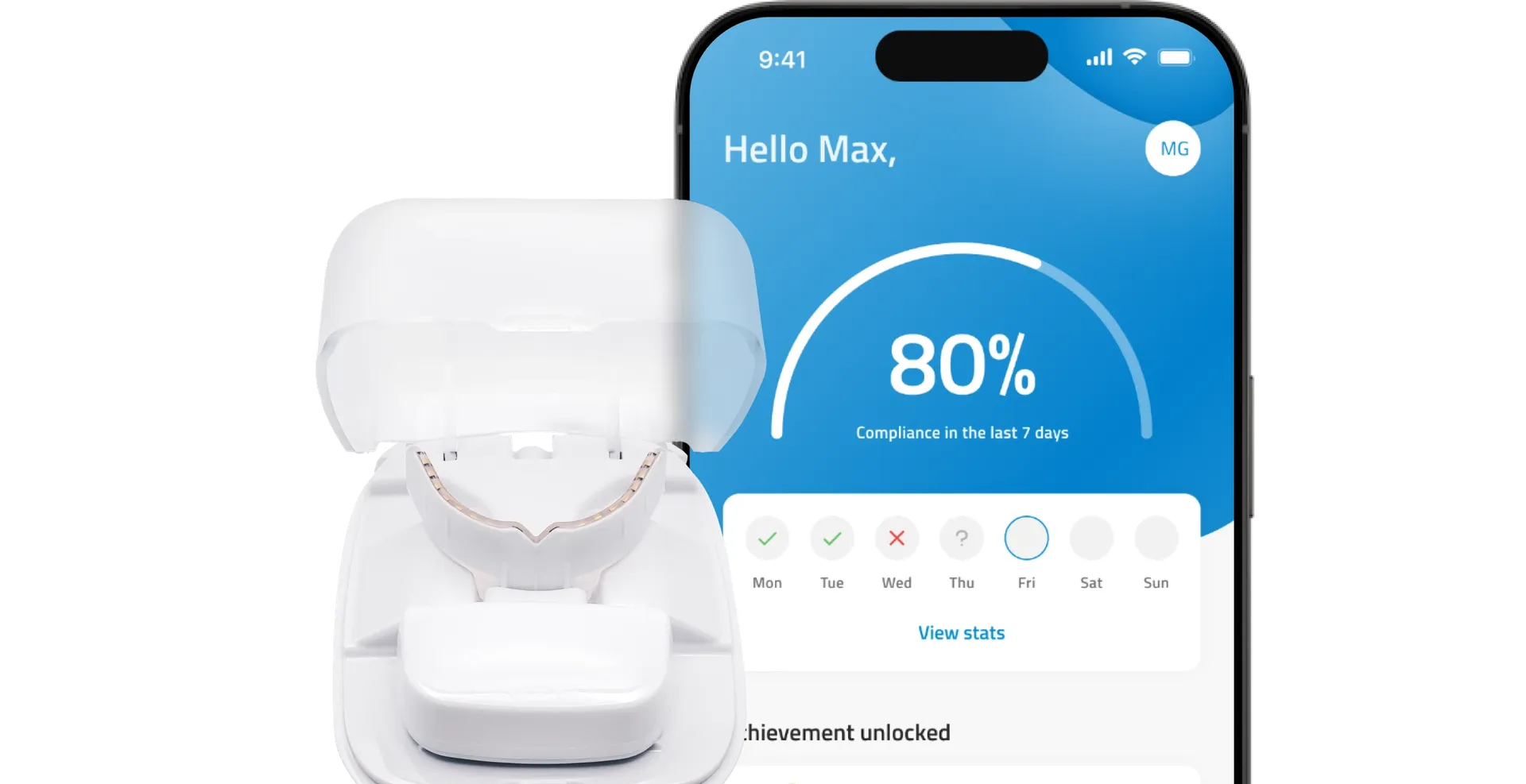
A beautiful smile in half the time
OrthoPulse® is the first and only approved medical device that uses near infrared light technology to shorten treatment time and reduce pain associated with orthodontic treatment.
Designed for use with braces or clear aligners.
Safe and simple. Results with just 10 minutes a day.
Helps your teeth align more precisely with your aligners.
US FDA cleared and EU MDR approved.

Time travel for your teeth
OrthoPulse® is an established device that uses near infrared light technology, also known as photobiomodulation, to stimulate the bone surrounding the roots of your teeth and facilitate tooth movement which may reduce treatment time for braces or clear aligners
Smart Digital Platform
- How It Works
Patients
OrthoPulse® offers a device that uses near infrared light to speed up orthodontic treatment, paired with a mobile app for tracking progress and reminders.
- For Doctors
Doctors
OrthoPulse® provides a doctor portal with real-time data and tools to monitor and manage patient treatments more effectively

More than 2,000,000 teeth moved faster and more predictably
Doctors around the world have had great success prescribing OrthoPulse® to accelerate treatment times.


We’d love to hear from you
Do you have any questions? Our team is here to help!